- Boron [B] – Element Details, Facts, Properties, Trends, Uses, Comparison with other elements
- Boron
- History of Boron
- How to Locate Boron on Periodic Table
- Boron Facts
- Boron Atomic Structure and Orbital Properties
- Ground State Electronic Configuration of Boron - neutral Boron atom
- Atomic Spectrum of Boron
Boron [B] – Element Details, Facts, Properties, Trends, Uses, Comparison with other elements

Boron
Boron is the 5th Element of Periodic table with atomic number 5, atomic weight 10.811. Boron, symbol B, has a Simple Trigonal structure and Black color. Boron is a metalloid element. Trivial name of Boron is triels, icosagens. Know everything about Boron Facts, Physical Properties, Chemical Properties, Electronic configuration, Atomic and Crystal Structure.

History of Boron
The element Boron was discovered by Joseph Louis Gay-Lussac in year 1808 in France and United Kingdom . Boron derived its name from borax, a minera.
Boron is a metalloid chemical element with symbol B and atomic number 5. Produced entirely by cosmic ray spallation and supernovae and not by stellar nucleosynthesis, it is a low-abundance element in both the Solar system and the Earth’s crust. Boron is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals
How to Locate Boron on Periodic Table
Periodic table is arranged by atomic number, number of protons in the nucleus which is same as number of electrons. The atomic number increases from left to right. Periodic table starts at top left ( Atomic number 1) and ends at bottom right (atomic number 118). Therefore you can directly look for atomic number 4 to find Beryllium on periodic table.
Another way to read periodic table and locate an element is by using group number (column) and period number (row). To locate Beryllium on periodic table look for cross section of group 2 and period 2 in the modern periodic table.
Boron Facts
Boron Atomic Structure and Orbital Properties
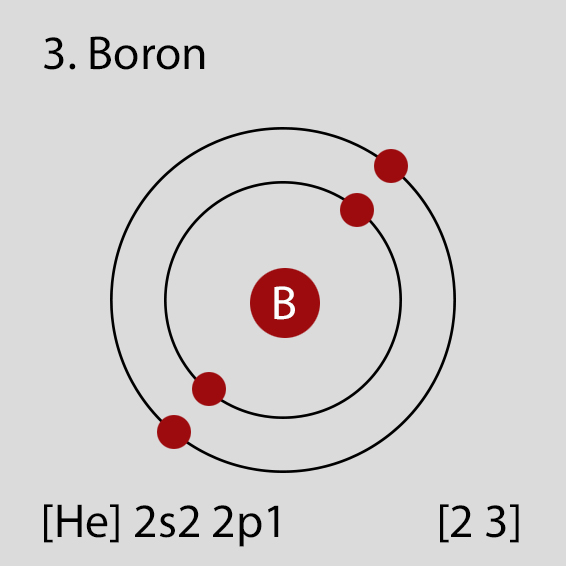
Boron atoms have 4 electrons and the electronic shell structure is [2, 2] with Atomic Term Symbol (Quantum Numbers) 1S0.
Properties
Atomic Structure of Boron
Ground State Electronic Configuration of Boron - neutral Boron atom
The ground state electronic configuration of Neutral Boron atom is [He] 2s2 2p1. The portion of Boron configuration that is equivalent to the noble gas of the preceding period, is abbreviated as [He]. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used.This is important as it is the Valence electrons 2s2 2p1, electrons in the outermost shell that determine the chemical properties of the element.
Unabbreviated electronic configuration of neutral Boron
Complete ground state electronic configuration for the Boron atom, Unabbreviated electronic configuration
1s2 2s2 2p1
Atomic Spectrum of Boron
